|
By: Sadia Akbar 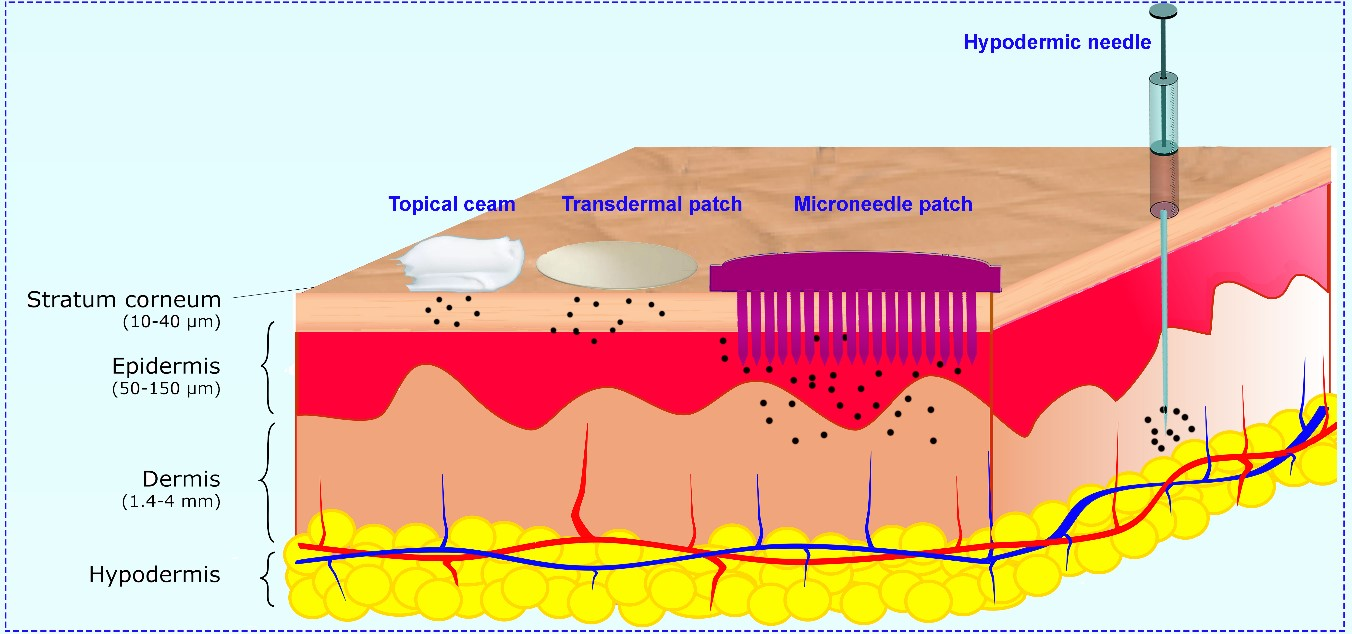 Attribution: Rezvan Jamaledin, Concetta Di Natale, Valentina Onesto, Zahra Baghban Taraghdari, Ehsan Nazarzadeh Zare, Pooyan Makvandi, Raffaele Vecchione, Paolo Antonio Netti, CC BY 4.0 Attribution: Rezvan Jamaledin, Concetta Di Natale, Valentina Onesto, Zahra Baghban Taraghdari, Ehsan Nazarzadeh Zare, Pooyan Makvandi, Raffaele Vecchione, Paolo Antonio Netti, CC BY 4.0 For anyone with a fear of needles, the prospect of a painless alternative is sure to be welcome. Microneedles are an emerging, cutting-edge form of drug delivery. The device consists of many microscopic needles, small enough to be measured in millionths of a metre, arranged on a patch. For further insight, each needle is about the depth of a sheet of paper and as wide as a strand of human hair. There are a variety of reasons why microneedles are a promising innovation in drug delivery. Being as small as they are, they pierce the outermost layer of skin to create micron size pathways to the epidermis but do not reach the pain receptors in the underlying dermis, and only scarcely penetrate the blood vessels. As a result, they are virtually painless. The sensation is said to be similar to Velcro against the skin–far more preferable than the sharp pinch of a traditional needle. Painlessness is an obvious advantage over traditional needles, but microneedles reign supreme over other widely used methods of transdermal drug administration like topical creams and transdermal patches as well. The therapeutic action of drugs delivered through these methods is limited by a layer of the skin called the stratum corneum, which acts as a semi-permeable barrier by only permitting certain molecules from passing through. With microneedles, the pathway through the epidermis allows the drug to enter systemic circulation directly, bypassing the stratum corneum, resulting in a greater effect. Perhaps the most promising application of microneedles is regarding the delivery of vaccines. Traditional liquid vaccines need to be kept refrigerated from the time they are manufactured to the time they are delivered in order to remain stable, a phenomenon known as the “cold chain”. Liquid vaccines can become inert, meaning inactive, if not kept at an adequate temperature, and a large proportion of vaccines produced are wasted for this exact reason. In fact, this cold chain has been presenting a challenge for the current delivery of COVID-19 vaccines. In contrast, microneedles can be prepared in a dry state, which means they work and are stored at room temperature, and do not require refrigeration. As such, they can greatly reduce the wasting of vaccines due to the cold chain and also allow for easier transport and cheaper production. Numerous microneedle vaccines are already in use, with many more undergoing clinical trials for use in diabetes, cancer, and neuropathic pain treatments. Researchers at Swansea University in Wales are even in the early stages of developing a microneedle vaccine for COVID-19, and are aiming to have a prototype produced by March 2021, following which they will advance towards clinical trials. Besides delivering vaccines, the use of microneedles has been explored for a variety of other applications. They may be used to painlessly draw blood for diagnostic testing, as the pressure change from making the punctures can force the blood into a collection device. A blood collection system using microneedles called TAP has already been approved in the U.S. and Europe and allows people to collect small samples of their own blood to later send to a lab or be used for self-monitoring. Microneedles can even be attached to biosensors, which are devices that can detect and convert biological reactions into electrical signals that can then be measured, allowing for the rapid measurement of glucose, cholesterol, immune cells, or other biological markers indicative of health. They are also being integrated with wireless communication devices to measure certain biological molecules, and this measurement is then used to determine and subsequently deliver the required dosages of a drug. This is a major advancement in the realm of personalized medicine and self-management of symptoms. Other benefits of microneedles include the reduced risk of transmitting blood-borne viruses, the reduced amount of hazardous waste from disposing of traditional needles, and their potential to be distributed in underserved areas due to the lack of costly equipment and training needed to administer them. Inevitably, microneedles are not devoid of limitations. They are not always sufficient in administering drugs that are needed in large doses, not all drugs are able to be administered through microneedles, and not all biological markers can be sampled using them. They can also present challenges with sensitive skin, such as irritation or allergic reactions. Several issues can also arise from their small size as the tips of the needles can break and potentially remain in the skin. However, these problems are very rare and likely to be overcome with more research and further development. Overall, being painless along with a host of other benefits suggests microneedles and research on them are bound to significantly expand and they are sure to be on the rise in the field of drug delivery and potentially revolutionize the industry. References: O'Day, E. (2020, November 10). Microneedles Could Enable Painless Injections and Blood Draws. Scientific American. https://www.scientificamerican.com/article/microneedles-could-enable-painless-injections-and-blood-draws/. Price, S. (2021, January 7). World first COVID-19 smart vaccine patch. Health Europa. https://www.healtheuropa.eu/world-first-covid-19-smart-vaccine-patch/104768/. Quinn, H. (2020, March 10). Explainer: what are microneedles and why do we need them? The Conversation. https://theconversation.com/explainer-what-are-microneedles-and-why-do-we-need-them-54623. Saey, T. H. (2021, January 30). Here's why COVID-19 vaccines like Pfizer's need to be kept so cold. Science News. https://www.sciencenews.org/article/coronavirus-covid-19-why-vaccines-cold-freeze-pfizer-moderna. Tothill, I. E., & Turner, A. P. F. (2015). Biosensor - an overview. ScienceDirect. https://www.sciencedirect.com/topics/nursing-and-health-professions/biosensor. Waghule, T., Singhvi, G., Dubey, S. K., Pandey, M. M., Gupta, G., Singh, M., & Dua, K. (2019). Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomedicine & Pharmacotherapy, 109, 1249–1258. https://doi.org/https://doi.org/10.1016/j.biopha.2018.10.078
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Categories |


 RSS Feed
RSS Feed